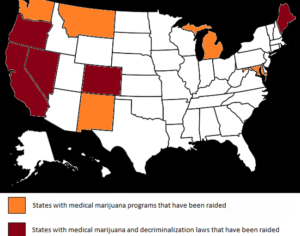
DEA continues to make empty promises on clinical cannabis research
Surprised? You shouldn’t be. The DEA possesses an abysmal track record when it comes to enabling scientists to engage in cannabis-specific studies in the United States, in particular research evaluating the plant’s therapeutic efficacy.
With such a checkered past, it is easy to see why many remain incredulous that the DEA’s latest actions are in good faith. And a closer look at the new rules provides even more reasons to be skeptical. For one thing, the proposed changes are woefully impractical. For another, they greatly expand the agency’s power and control over decisions that ought to be best left up to health experts and scientists. Specifically, the new rules continue to grant the DEA sole discretion to decide which applicants, if any, will be permitted to grow cannabis for research purposes, and it provides no timeline under which the agency must act on the 30+ applications already before it. Further, the proposed rules mandate the agency to become the sole purchaser of all cannabis produced by federally licensed entities. Yet, nowhere do the rules mandate that the agency pay fair market value for these products, or do they ensure that the DEA purchases them at a price that allows manufacturers to cover their start-up costs — which will no doubt be enormous — or turn a profit.
Original Article (The Hill):
DEA continues to make empty promises on clinical cannabis research
Artwork Fair Use: J. Patrick Bedell












































































































































































































































































































































Recent Comments