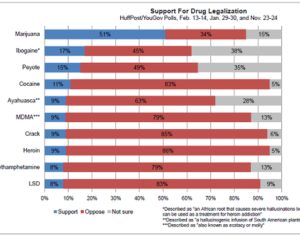
In a first, FDA orders recall of a ‘contaminated food’…
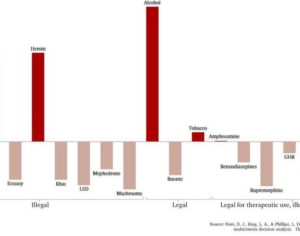
There is no reason for DEA [or FDA] to schedule Kratom, and it would be criminal to leave millions of Kratom users, and opiate-dependent populations, with no alternatives to pain and suffering, other than a return to increased use of prescription opioids or resorting to illegal purchase on the black market. -Jane Babin, Ph.D., Esq. University of San Diego School of Law, J.D. Purdue University, Ph.D., Molecular Biology
The mandatory recall was the agency’s first-ever recall of a contaminated food item. In general, dietary supplements are considered foods under federal law. In the case of kratom, a botanical from Southeast Asia, FDA officials determine whether the products are a food or an unapproved drug depending on the way they are labeled.
The FDA doesn’t have the authority to order mandatory recalls for drugs and seeks voluntary recalls if it sees a safety problem. But it got mandatory-recall authority for tainted food under the Food Safety and Modernization Act that went into effect in 2011. “This action is based on the imminent health risk posed by the contamination of this product with salmonella, and the refusal of this company to voluntarily act to protect its customers and issue a recall, despite our repeated requests and actions,” FDA Commissioner Scott Gottlieb said in a statement … In this instance, two samples of kratom products manufactured by Triangle Pharmanaturals of Las Vegas, Nevada, sold through … [a retail location] in Tigard, Oregon and collected by the Oregon Public Health Division, tested positive for salmonella, as did four additional samples of various types of kratom product associated with the firm collected by the FDA.
Original Article (Washington Post):
In a first, FDA orders recall of a ‘contaminated food’ & FDA News Room
Artwork Fair Use: gnokii (Open Clipart)