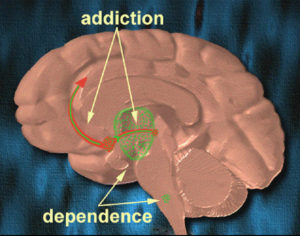
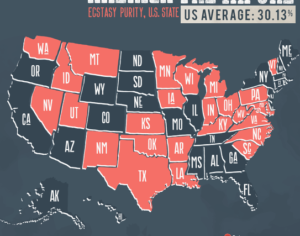
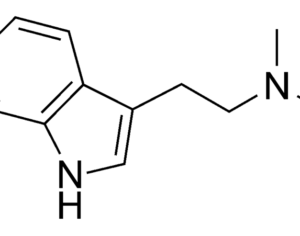
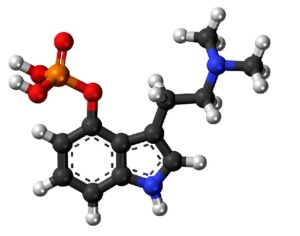
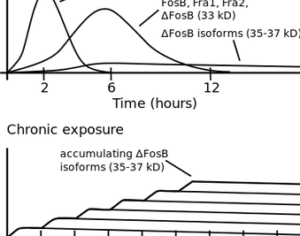
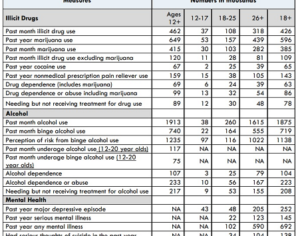
Hallucinogens: future of mental health treatment?
“Most powerful substances that we know of, that have powerful effects on the central nervous system, are like any powerful tool,” says Johnson, who has studied how psilocybin affects depression. “They can have dangerous effects, or beneficial effects, if judiciously used in a context where the dangers are known and mechanisms are in place to address them.”
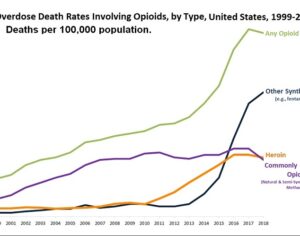
“You have an entire array of institutions that need to sign off — the FDA; the DEA; at a university, the institutional review board,” Johnson says. “There are a lot of hoops to jump through.” The FDA allows Schedule I drugs, which include marijuana, to be used for clinical research under a process known as an Investigational New Drug application, agency spokeswoman Sandy Walsh says. When a study gets approved, the drugs involved “are subject to the most stringent restrictions” and have to be stored and kept track of under rules approved by the DEA. DEA spokesman Russ Baer says anyone who wants to study a Schedule I drug has to submit their proposed research protocols to the agency. Everyone involved in handling the drug has to be registered with the DEA, which judges whether the study plans will keep the drugs under control. “Since we’re not scientists, we’re not evaluating the veracity of the scientific research project,” Baer says — that’s the job of the FDA and any other research agencies involved. “What we’re required to do is to make sure there are appropriate safeguards in place to ensure security protocols are maintained, to make sure any controlled substance is not subject to theft or diversion from legitimate science on one hand to the black market on the other.”…There are more than 400 research projects now underway involving marijuana alone, Baer says, and he’s not aware of any drug study the agency has rejected or halted. In addition, he said the agency has been having talks with scientists about streamlining the agency’s approval process. “We support scientific research. We want to remove any sort of bureaucratic hurdles that may have existed in the past,” Baer says.
Original Article (WebMD):
Hallucinogens: Future of Mental Health Treatment?
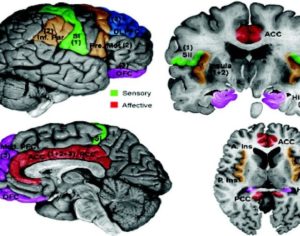
Artwork Fair Use: Mikael Häggström