FDA breakthrough therapy status to LSD analog…
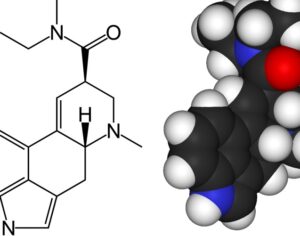
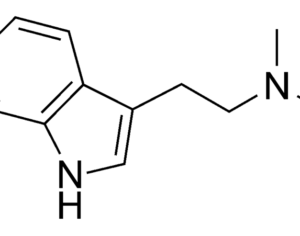
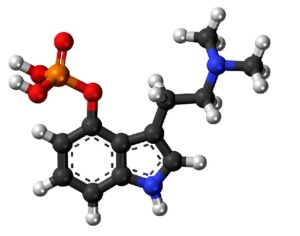
…an LSD analog, L S D (Lysergide D-Tartrate) was given “breakthrough therapy” designation by the FDA for treating anxiety.
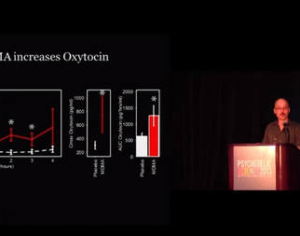
…this analog… a tartrate salt form… called MM-120. A single oral administration (100 µg) of this psychelic analog, MM120, maintained a clinically and statistically significant reduction in symptoms compared to placebo at 12 weeks — with a 65% clinical response rate and 48% clinical remission rate. Contrasting to most research with psychedelic medicine, this trial didn’t include psychotherapeutic assistance as part of its protocol.
Original Article (Instagram):
Shulgin Foundation
Artwork Fair Use: Matthew Woitunski




























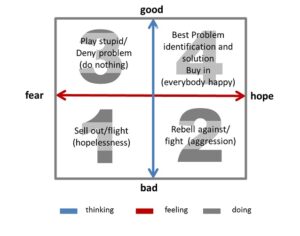
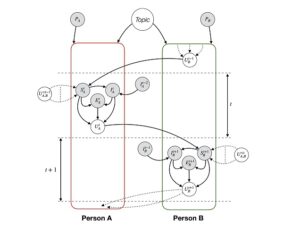


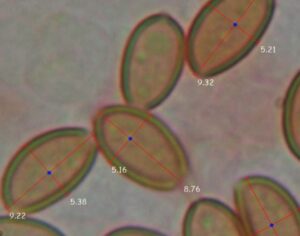






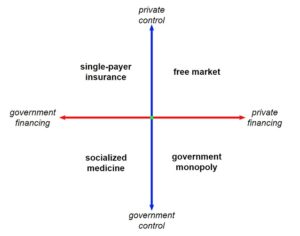

















































































































































































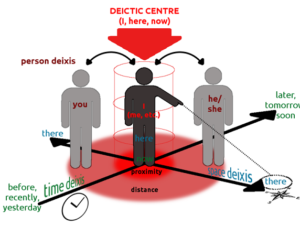
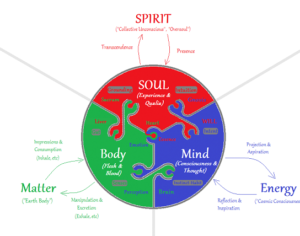



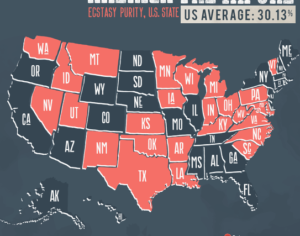



































































































Recent Comments