FDA Chief Scott Gottlieb abruptly resigns
Gottlieb’s FDA has earned its fair share of criticism.
Gottlieb also took fire for his agency’s decision to approve Dsuvia, the tablet form of an opioid painkiller more potent than fentanyl. He was also criticized for pushing back the timetable for when new regulatory rules would apply to e-cigarette products, similar to how tobacco cigarettes are regulated. Initially, the process would have started in 2018, but Gottlieb delayed it until 2022—a decision that prompted a lawsuit against the agency by several major public health groups, including the American Heart Association. [Numerous scientific experts also took stock with apparently misleading evidence regarding the safety of whole plant botanical kratom and evidence which apparently was generated under the supervision of Gottlieb].
Original Article (Gizmodo):
FDA Chief Scott Gottlieb abruptly resigns
Artwork Fair Use: G41rn8











































































































































































































































































































































































































































































































































































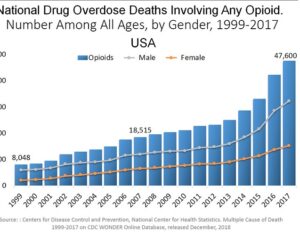
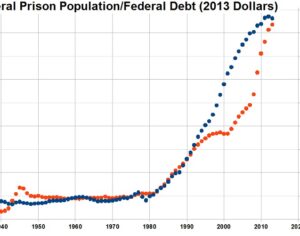

























































































































Recent Comments