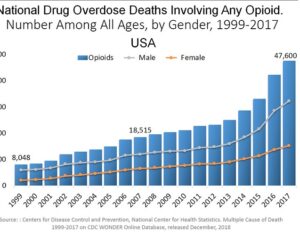
DEA to increase psilocybin… production for research purposes
The United States Drug Enforcement Administration (“DEA”) has proposed an increase in the manufacture… psilocin and psilocybin for clinical trial and research purposes… stated that its objective was to assist in the development of new federally approved therapies.
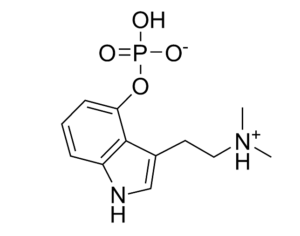
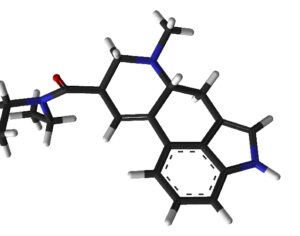
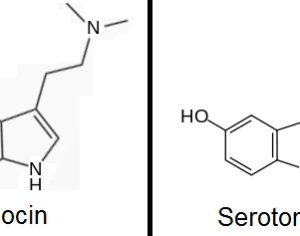
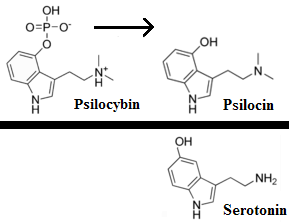
…increased its production goals for psilocin and psilocybin, which are the main psychoactive components of magic mushrooms. Initially, the quota for psilocybin was set at 50 grams. This has been adjusted to 1,500 grams, which is roughly a 2,900% increase. The original quota for psilocin was also 50 grams, with the DEA now proposing this amount be increased to 1000 grams. [Interested persons may file written comments on this notice in accordance with 21 CFR 1303.13(c) and 1315.13(d). To ensure proper handling of comments, please reference “Docket No. DEA-688A” on all correspondence, including any attachments. Electronic comments must be submitted, and written comments must be postmarked, on or before October 4, 2021… Based on comments received in response to this notice, the Administrator may hold a public hearing on one or more issues raised. Please note that all comments received in response to this docket are considered part of the public record. They will, unless reasonable cause is given, be made available by the Drug Enforcement Administration (DEA) for public inspection online at http://www.regulations.gov. Such information includes personal identifying information (such as your name, address, etc.) voluntarily submitted by the commenter.]
Original Article (Psychedelic Newswire & Federal Register):
DEA to increase psilocybin, cannabis production for research purposes & Proposed adjustments to the aggregate production quotas for schedule I and II controlled substances and assessment of annual needs for the list I chemicals ephedrine, pseudoephedrine, and phenylpropanolamine for 2021
Artwork Fair Use: Public Domain















































































































































































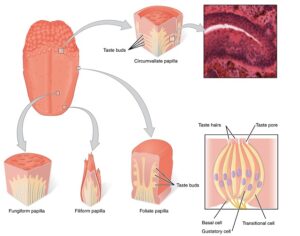


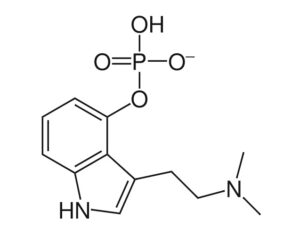



















































































































































































































































































































































































































































































































Recent Comments