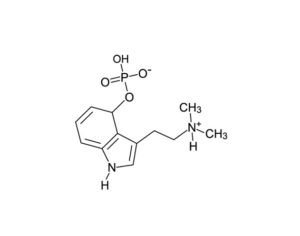
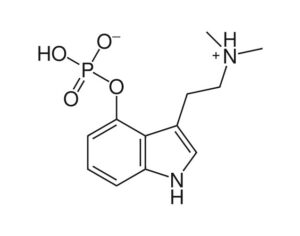
The psychedelic research hiatus and its relevance today
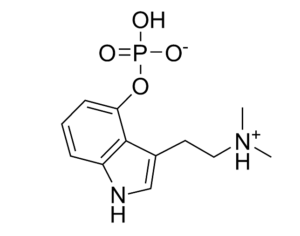
[In the 1950’s and 60’s] psychedelics were of little interest to pharmaceutical companies as the intervention seemed to treat illness, rather than alleviate symptoms. Consequently, the financial imperatives were lacking; fully recovered patients would not require continued medication… Sandoz was reluctant to share such proprietary and patented data (Bonson, 2018). When required by the FDA, the company only provided very limited animal toxicity data to prove safety. Sandoz never conducted trials to properly assess the safety and efficacy of LSD. In the 1950s, Sandoz ignored FDAs request to submit an application that would constitute a first step towards the formal registration of LSD as an approved drug. In 1963… regulations had caused scientists who wished to conduct research with LSD to be forced to provide data on its chemistry and manufacturing details to the FDA… The patent for LSD expired in the 1960s, certainly making it even less appealing for pharmaceutical companies at that point in time.
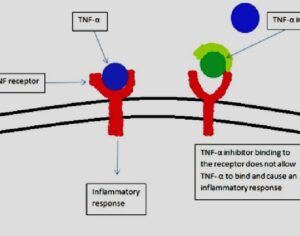
The golden standard of clinical drug research — randomized controlled trial (RCT), became standardised. The standard was targeted at taking the direct biological actions of drugs into account for evaluation. All other factors constituted biases to be eliminated. (Oram, 2018). RCT however, is fundamentally incompatible with the psychedelic treatment. LSD is perceived as lacking inherent therapeutic effects, but rather displaying therapeutic effects through its ‘extra-pharmacological’ effects (the experiences) it exerts. Besides, the RCT model assumes that neither the research subjects nor the researchers are able to determine if the drug or placebo has been administered. Naturally, this constitutes a major challenge with regards to psychedelics.
Original Article (Medium):
The psychedelic research hiatus and its relevance today
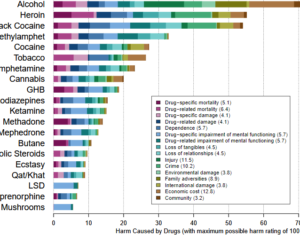
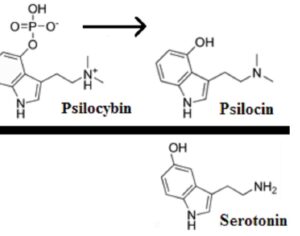
Artwork Fair Use: Thomas Shafee

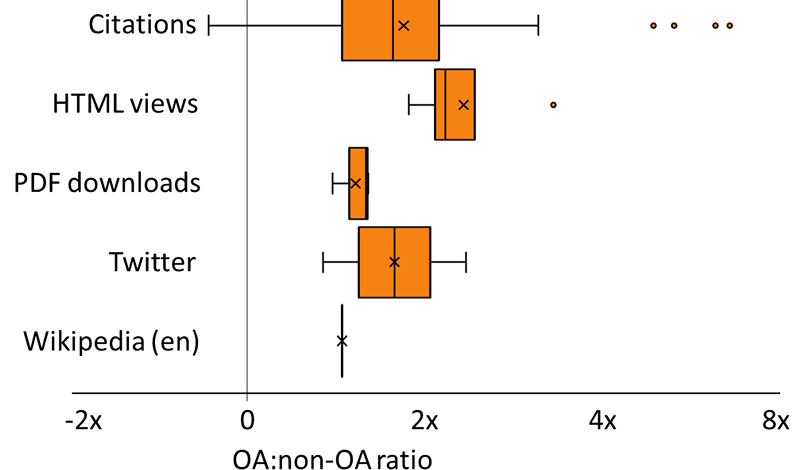









































































































































































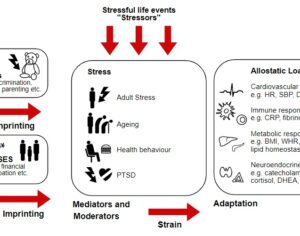










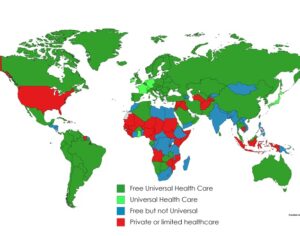




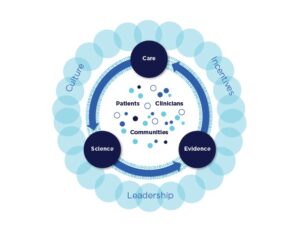




































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































Recent Comments