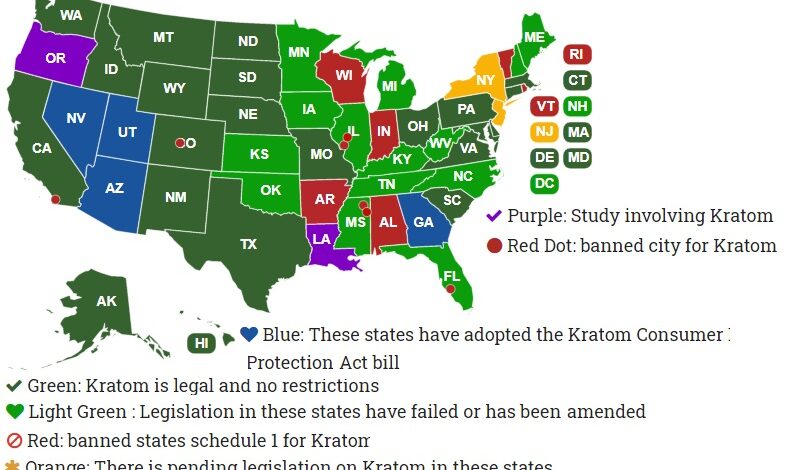
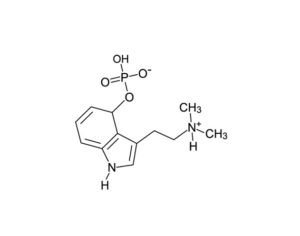
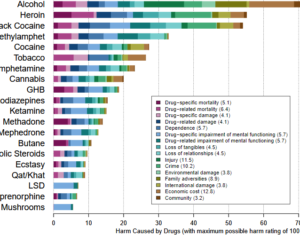
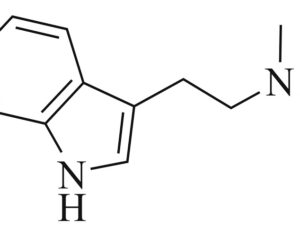
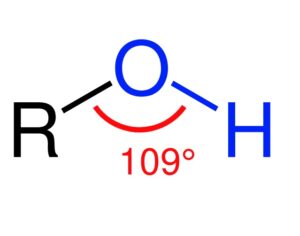
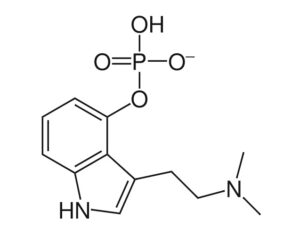
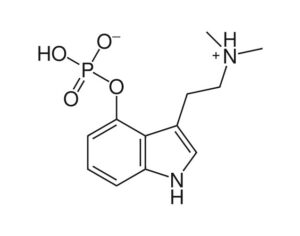
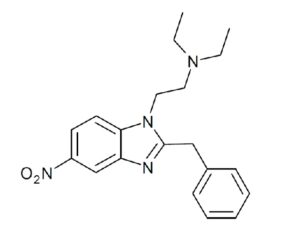
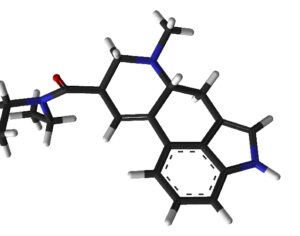
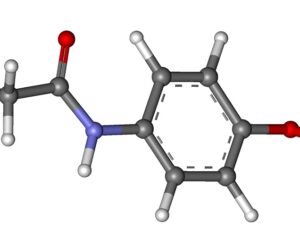
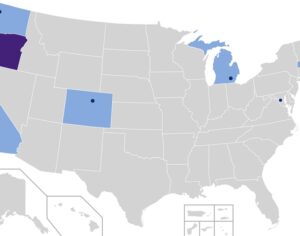
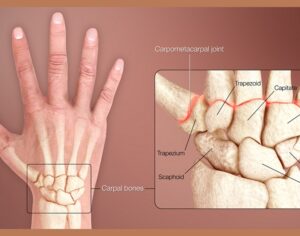
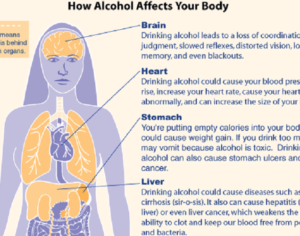
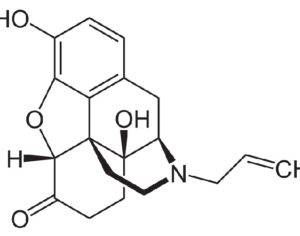
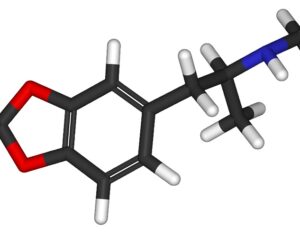
Ohio plans to ban kratom…
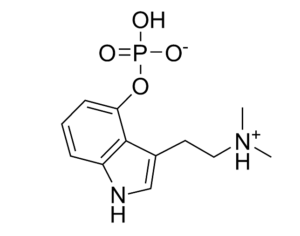
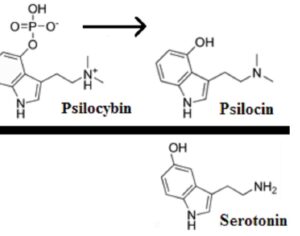
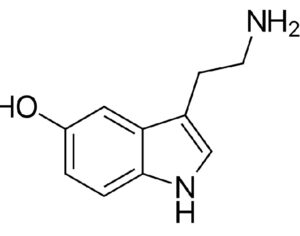
These studies demonstrate kratom’s potential medical uses. By classifying the active compounds in kratom as Schedule I substances, Ohio’s Board of Pharmacy would prevent further medical research into its benefits (similar to the obstacles the DEA has imposed on research of marijuana, a Schedule I substance).
At the time of the assessment’s release, FDA Commissioner Scott Gottlieb stated that “there is no reliable evidence to support the use of kratom as a treatment for opioid use disorder.” Yet the Ohio Board of Pharmacy’s own assessment of kratom reveals its potential as a medicine: “People addicted to heroin,” reads the report, “use [kratom] to alleviate opiate withdrawal symptoms.”
DEEP DIVE
“Ironically, the greatest danger of Kratom these days is from fake products contaminated with substances approved by the FDA.”
-From the Article Kratom : the CBD (Cannabadiol] of the Opiod World?
”[Even worse] the FDA weaponizes ‘opioid’ label against kratom consumers”
-From the Article FDA Weaponizes opiod label against Kratom Consumers
The FDA’s analysis (announced on Feb. 6th, 2018) was “an order of magnitude less rigorous than” many of the studies that had already been conducted on kratom.”
-From the Article FDA releases [alleged] kratom death data, undermines its own claims…
Original Article (Filtermag):
Ohio plans to ban kratom…
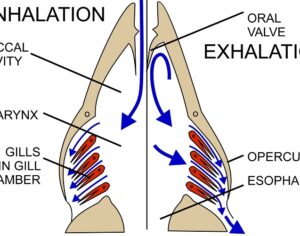
Artwork Fair Use: Speciosa