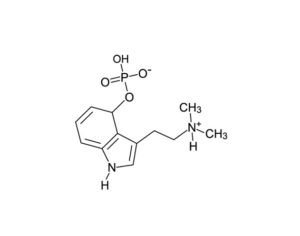
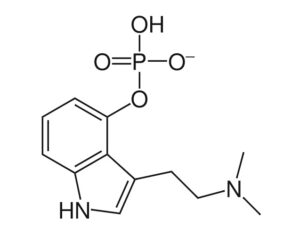
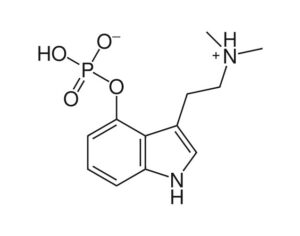
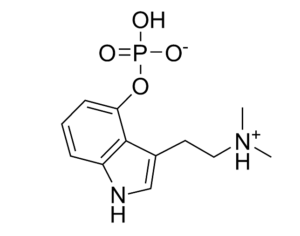
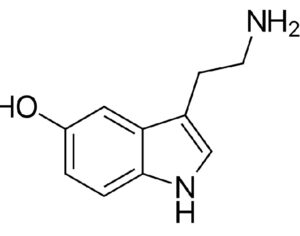
Psilocybin receives [Non-Profit] ‘breakthrough therapy’ designation from the FDA
“What is truly groundbreaking is FDA’s rightful acknowledgement that MDD (Major Depressive Disorder], not just the much smaller treatment-resistant depression population, represents an unmet medical need and that the available data suggest that psilocybin may offer a substantial clinical improvement over existing therapies.” -Dr. Charles Raison, MD
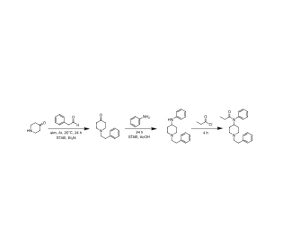
Compass Pathways, a pharmaceutical research firm, received the designation for studying psilocybin as a treatment for treatment-resistant depression.
Original Article (Business Wire):
FDA grants breakthrough therapy designation to Usona Institute’s psilocybin program for major depressive disorder
Artwork Fair Use: Non profit organization lawyers oakland











































































































































































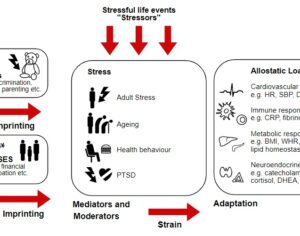










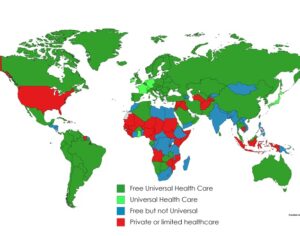




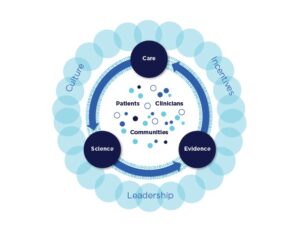




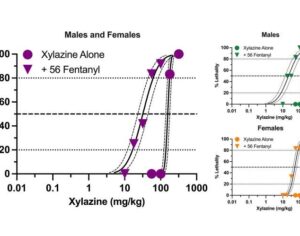













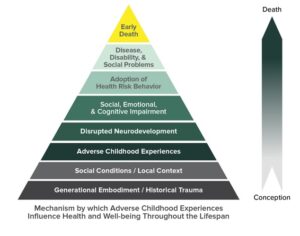





































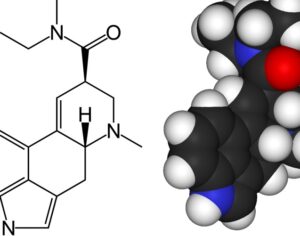




























































































































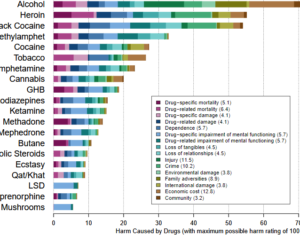




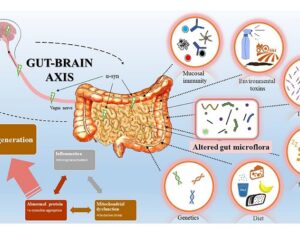

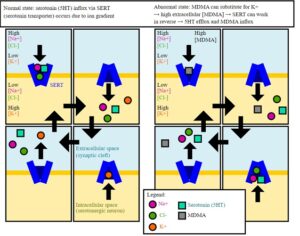




































































































































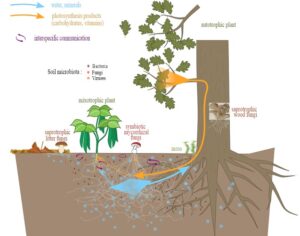







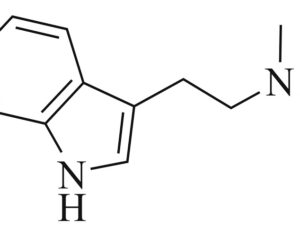































































































































































































































































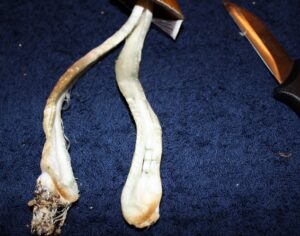
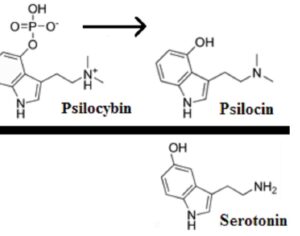































































































































































































































































































































































































































































































































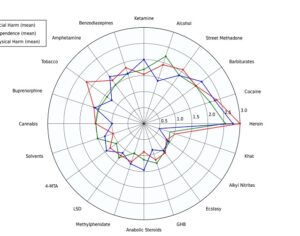


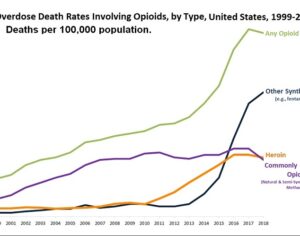












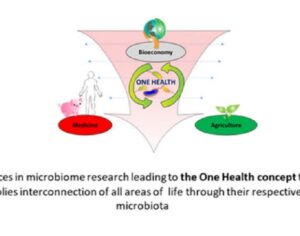




















































































































































































































































































































































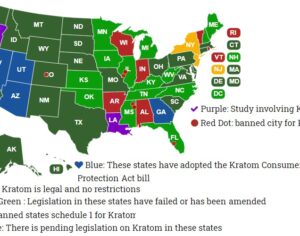

























































































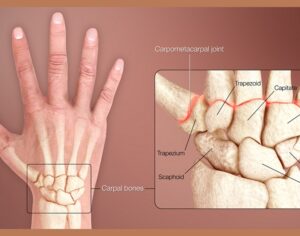




















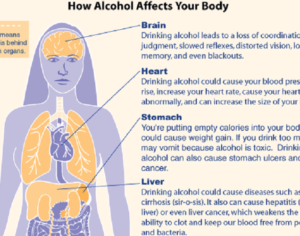




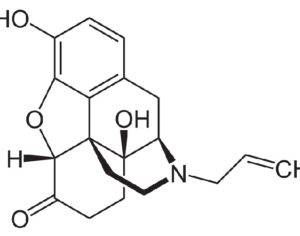



























































































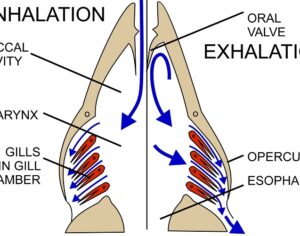







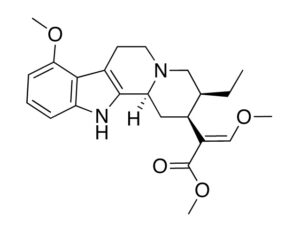














































































Recent Comments