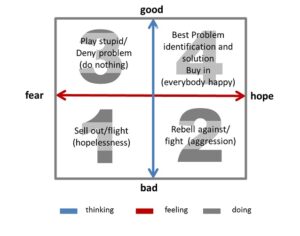
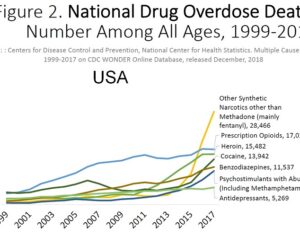
Far out, man: ATAI raised $43m to conduct psychedelic medicine research
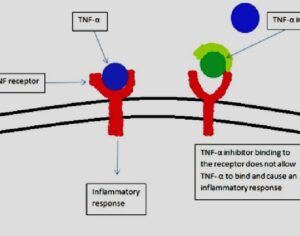
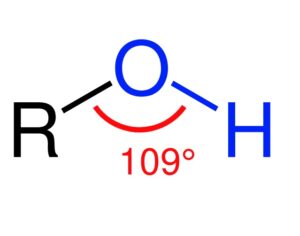
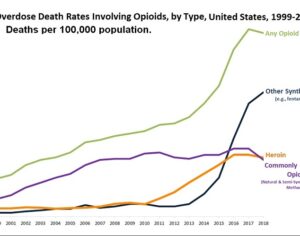
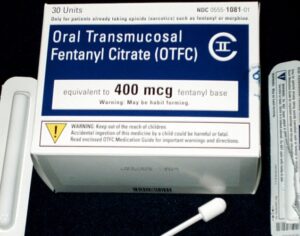
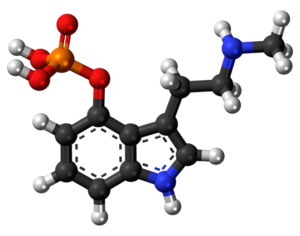
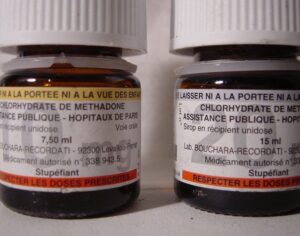
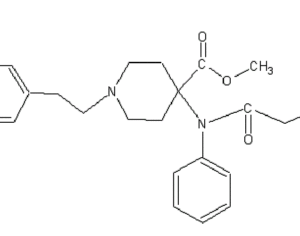
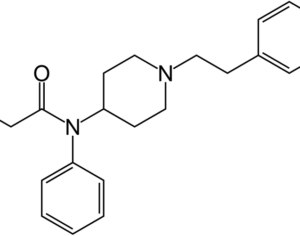
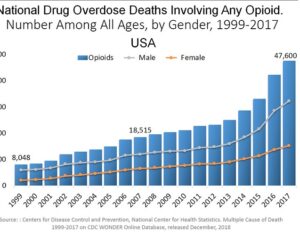
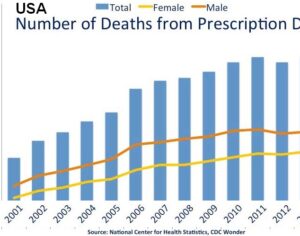
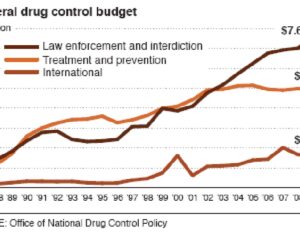
Weeks after the FDA approved Johnson & Johnson’s ketamine spray for depression [named esketamine, at a cost of $885.00 per mg vs. ketamine (generic), which is already available at a cost of $1.59 per 100mg)] a group of investors joined forces in the largest-ever private financing round for a psychedelic medicine biotech company.
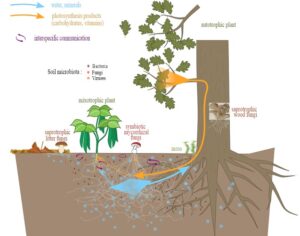
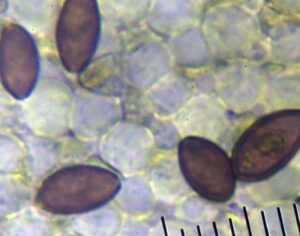
“We were not interested in what people learn about mushrooms from books, but what untutored country folk know from childhood — the folk legacy of the family circle,” Wasson recalled. “It turned out that we had happened on a novel field of inquiry.” Maria Sabina, however, had a different idea, “From the moment the foreigners arrived to search for God , the saint children lost their purity,” she said. “They lost their force; the foreigners spoiled them. From now on they won’t be any good. There’s no remedy for it.” Wasson, for his part, agreed. He expressed remorse for the rest of his life for his role in popularizing the… use of magic mushrooms. “A practice carried on in secret for three centuries or more…”
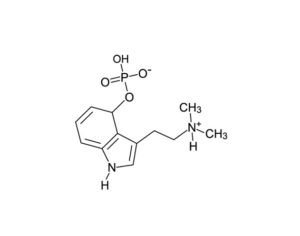
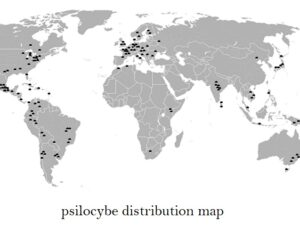
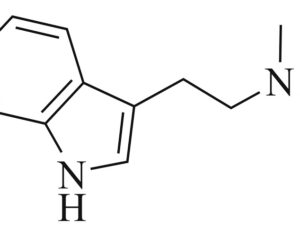
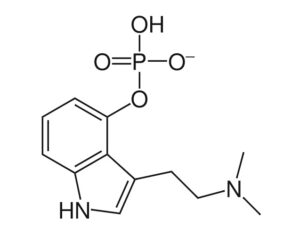
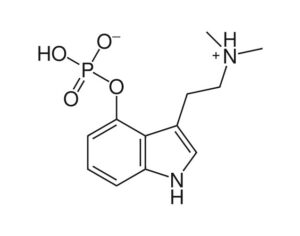
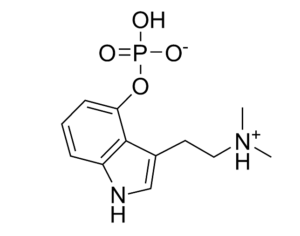
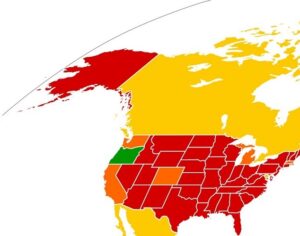
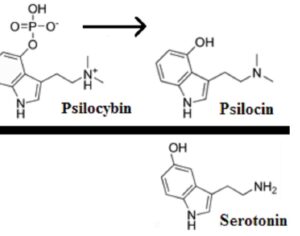
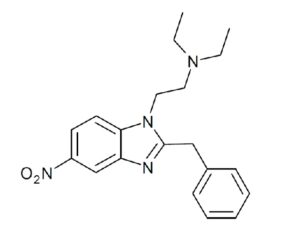
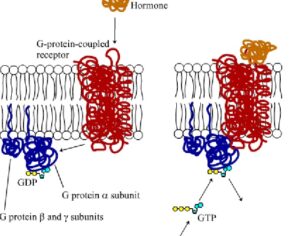
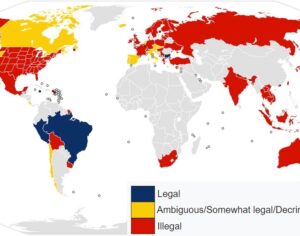
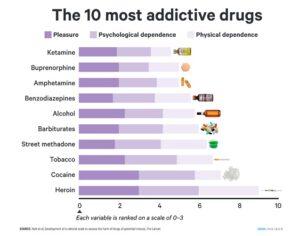
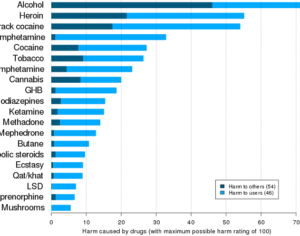
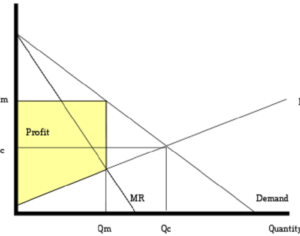
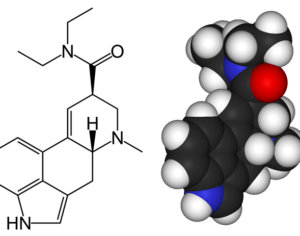
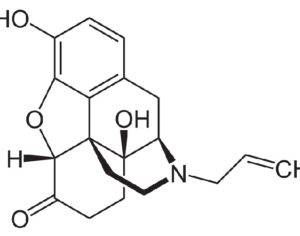
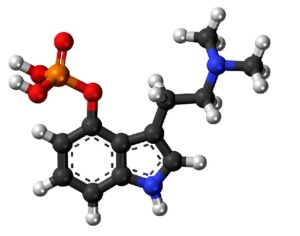
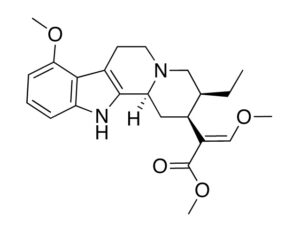
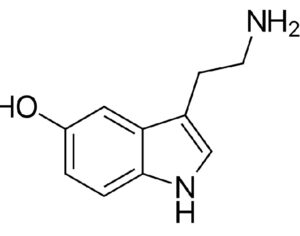
ATAI Life Sciences, a German biotech startup focused on depression and other mental health disorders, raised $43m in Series B funding. It is now the highest valued company in the space, valued at $240m. Recreational use of psychedelics became popular in the 1960s, but it didn’t take long for the US government to classify most of them as “drugs of abuse.”But, according to CNBC, recent clinical studies have found loads of evidence that some of the trippy dippys actually can help patients with certain mental illnesses. ATAI is currently funding clinical trials for “formerly stigmatized compounds,” like psilocybin (the activator in magic mushrooms) and arketamine (a different form of ketamine than what’s in J&J’s nasal spray). Psychedelics are having a moment: Not only are regulators peaking at the therapeutic potential, investors are down to follow the white rabbit as well — of course, that comes with good, bad, and ugly. The good: Higher exposure brings in more funding for research. The bad: Big Pharma is an atrocious industry, where most companies are more in it for the cash than for patient well-being. The ugly: Rising stars like Compass Pathways, which at one time seemed to be conducting psilocybin trials for the right reasons, sometimes take shady turns to ensure monopolization of a product. Oh, and guess which company is Compass Pathways’ largest investor? [That’s right, ATAI]. [The summary: While ATAI is currently funding clinical trials for their self-described “formerly stigmatized compounds,” like psilocybin (simply one of the psychoactive compounds in magic mushrooms)… some psychedelic historians and philosophers argue that while the “compounds” are increasingly de-stigmatized, the whole plant botanicals and fungi themselves remain so, hence, legislation crafted under the World Trade Organization and current start-up funding approaches reinforce a system which was designed for inventions which were “formal”, such as those carried out in universities and laboratories, or as part of industrial R&D. As of now, the WTO does not recognize technology innovations by farmers, artisans, or grassroots innovators, which happen in an informal setup. These form a large part of traditions knowledges [and are routinely ignored, neglected, and otherwise derided or seen as fiscally irrelevant, ignoring hundreds, if not thousands of years of ecologically sensitive and entourage/ensemble effect influenced practices.]
Original Article (The Hustle):
Far out, man: ATAI raised $43m to conduct psychedelic medicine research and Timeline
Artwork Fair Use: ECFES/Edelic Public Lending Library













































































































































































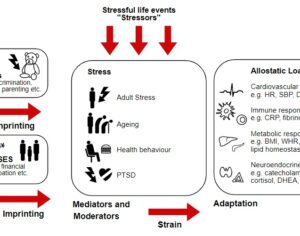










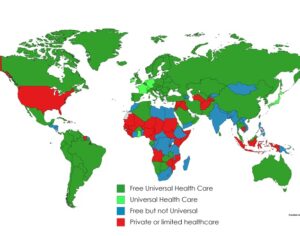




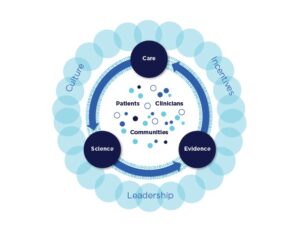




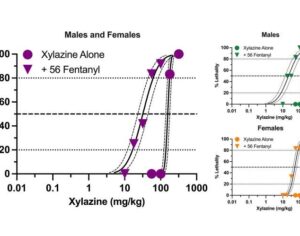













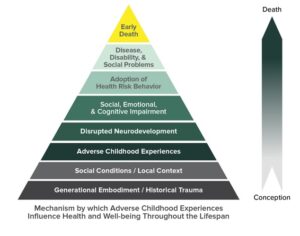





































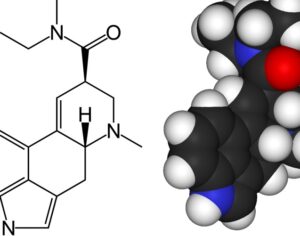





























































































































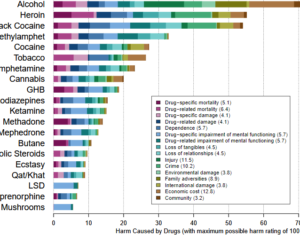




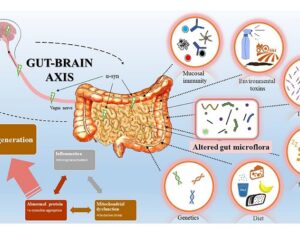

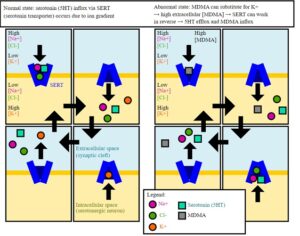





















































































































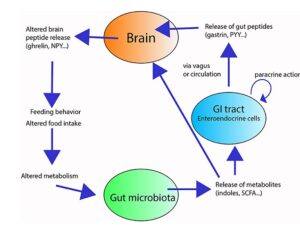




































































































































































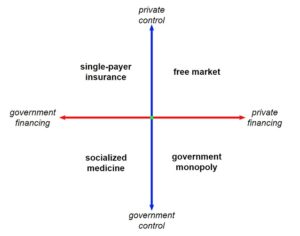






























































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































Recent Comments